 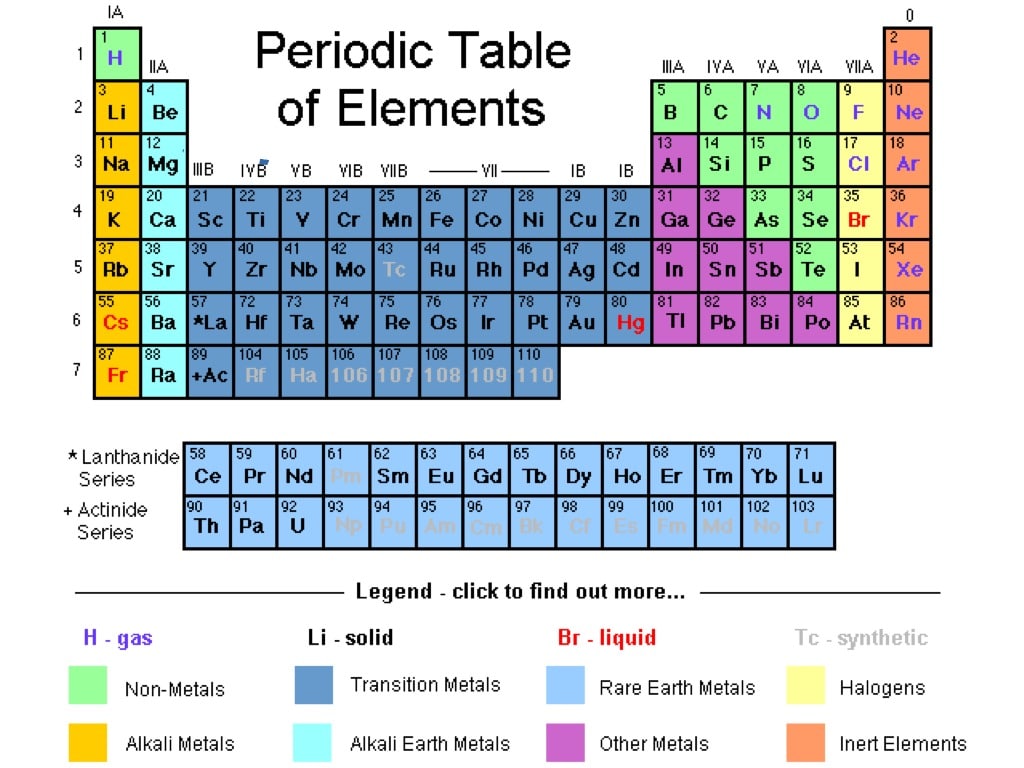
As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. 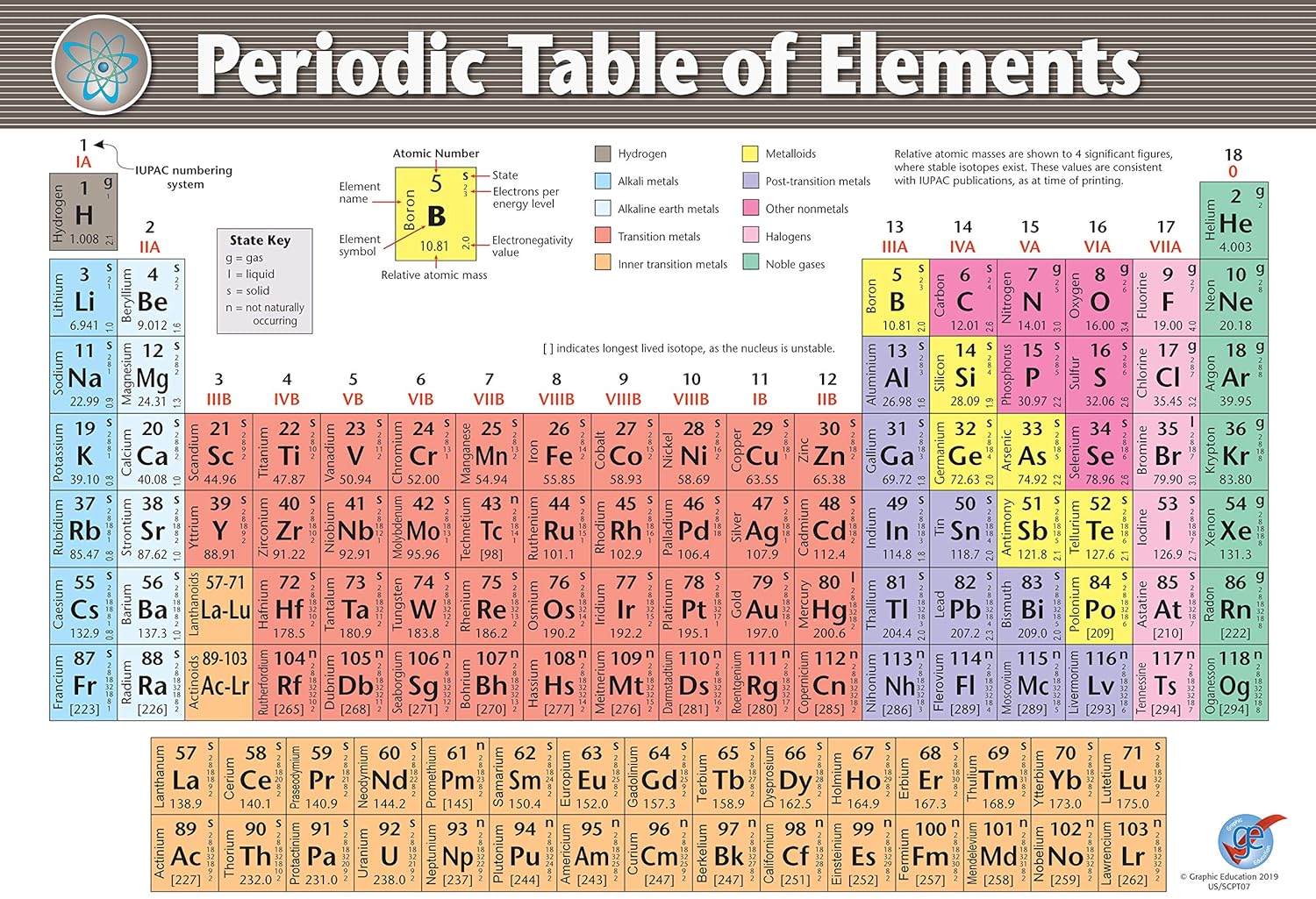
For example, students should be familiar with the layout and keywords associated with the periodic table. This includes: 1.1 Metals and non-metals 1.2 Groups and periods 1.3 The elements of Group 1 1.4 The elements of Group 7 1. result in more meaningful learning for all students. Element atomic number increases as you move down a group from top to bottom or across a period from left to right. Both groups and periods reflect the organization of electrons in atoms. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. Created for the Activate 2 Chapter 1: The Periodic Table topic.This bundle comes complete with fully differentiated and resourced lessons from the topic. A group is a vertical column down the periodic table, while a period is a horizontal row across the table. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. An interactive Periodic table can be found Periodic Table of the Elements, LibreTexts. The semimetals lie along a diagonal line separating the metals and nonmetals. The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. Using colored pencils, color each group on the table as follows. Your teacher will give you a copy of the periodic table to color. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. 1: The Periodic Table Showing the Elements in Order of Increasing Z. Part 1: This worksheet will help you understand how the periodic table is arranged. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
